India Pharma Outlook Team | Wednesday, 25 March 2026
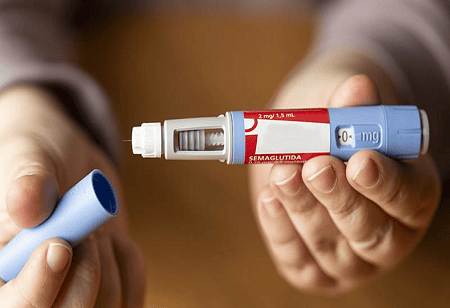
Zydus Lifesciences Limited has announced the launch of its Semaglutide injection in India under the brand names SEMAGLYN, MASHEMA, and ALTERME, following the expiry of the product’s patent.
The therapy has received approval from the Drug Controller General of India for the treatment of Type 2 Diabetes Mellitus and obesity, two of the country’s fastest-growing health concerns.
The new therapy comes with a reusable multi-dose pen device, offering a practical alternative to conventional single-use pens. Patients needing dose adjustments often require multiple pens with existing methods. The new device allows users to select varying dose strengths from a single pen, simplifying administration and improving adherence.
Also Read: How Digital Maturity is Powering Pharma's GMP Evolution
The Semaglutide injection will be available in a 15 mg/3 ml cartridge, manufactured at Zydus Biotech Park in Ahmedabad. The company has stated that the average monthly cost of treatment will be around Rs. 2,200, making it more affordable for patients requiring long-term therapy.
India faces a rising burden of metabolic disorders. According to the International Diabetes Federation, nearly 89 million adults in India are living with diabetes, representing over 10 percent of the adult population. Obesity prevalence has also surged, with rates among women increasing from 12.6 percent to 24 percent and among men from 9.3 percent to 22.9 percent in recent years. These trends highlight the urgent need for effective and cost-efficient treatment options.
GLP-1 based therapies are gaining recognition for managing both diabetes and obesity. With this launch, Zydus Lifesciences aims to enhance access to these therapies while improving patient convenience through device innovation.
Operating globally, Zydus Lifesciences is involved in the discovery, development, manufacturing, and marketing of a broad range of therapies. The group employs over 29,000 people worldwide, including a dedicated research team focused on advancing new treatments.