Thiruamuthan, Assistant Editor, India Pharma Outlook

The pharmaceutical sector in India is a rapidly evolving sector in the global markets, as it has ceased to be manufacturing-centered and the main focus now has shifted towards advanced therapies, complex generics and biologics. According to IBEF, India is the third biggest pharmaceutical manufacturer in the world, and it produces nearly 20 percent of all generic drugs that are consumed in the world. The nation is becoming part of critical role in global healthcare, as it is increasing its services in biosimilars, vaccines, and contract manufacturing services.
But how does this growth become a source of prowess? The answer to this is the industry that is supported by positive government policies, including the Production Linked Incentive (PLI) scheme and the Strengthening Pharmaceutical Industry (SPI) scheme, which would create an environment promoting innovation and making India more globally competitive.
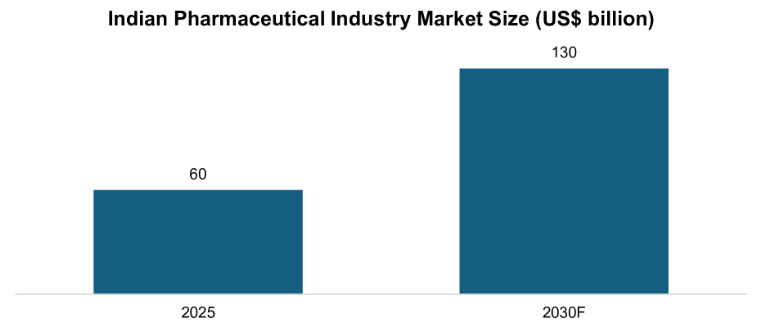
In the year 2024-25, pharma exports peaked at an all-time high of USD 30.47 billion, which represents a strong yearly growth of 9.4 percent, and the domestic market stands at USD 60 billion with a forecast of USD 130 billion in 2030. On paper, India has the third-largest volume globally in production and supplies approximately 20 percent of the generic drugs in the world. However, the country is highly reliant on imports of high value biologics and biosimilars, which are the essentials in the treatment of non-communicable diseases, including diabetes, cancer, and autoimmune diseases. The Indian biopharmaceutical market alone is estimated to be USD 8.95 billion in 2025, and to reach USD 17.41 billion USD in 2033, with a CAGR of 8.3 percent.
(Source: IBEF)
To bridge this gap and position India as a global biopharma innovation and manufacturing hub, the Union Budget 2026-27 announced the Biopharma SHAKTI Scheme (Strategy for Healthcare Advancement through Knowledge, Technology and Innovation) with a massive outlay of Rs 10,000 crore over five years (starting FY 2026-27). The scheme, formally detailed in a Press Information Bureau release on 10 March 2026, aims to build an end-to-end ecosystem for biologics, biosimilars, and advanced therapies while reducing regulatory timelines and fostering innovation.
This comprehensive guide explains the scheme’s objectives, eligibility, benefits, and—most importantly—how to prepare for and eventually apply once operational guidelines are notified.
Announced by Finance Minister Nirmala Sitharaman and elaborated by Minister of State for Chemicals and Fertilizers Anupriya Patel, Biopharma SHAKTI addresses India’s shifting disease burden toward non-communicable diseases. Biologics—complex medicines derived from living organisms—are the future of affordable, high-quality healthcare.
The scheme structures the already existing initiatives like the Production Linked Incentive (PLI) for Pharmaceuticals and the Scheme for Promotion of Research and Innovation in Pharma MedTech Sector (PRIP), but shifts focus decisively from generics to value-added biologics.
While detailed eligibility criteria will be outlined in upcoming operational guidelines, the scheme openly targets:
With this exceptional support from the Government of India, MSMEs stand out to benefit significantly in lower-barrier segments such as bioprocess equipment, nutrient media, and niche biosimilars. Even startups can leverage CDMO models to de-risk development. The main priority is expected for projects addressing non-communicable diseases and those aligned with “Make in India” and export goals.
Successful applicants can expect:
Many industry leaders have hailed and supported the scheme as a game-changer for moving India “from volume to value leadership.” With over USD 250 billion worth of biologics going off-patent globally in the coming decade, timely participation could unlock massive export opportunities.
Important Update (as of March 2026): Detailed operational guidelines, application portal, and call for proposals have not yet been notified by the Department of Pharmaceuticals (DoP). The scheme was announced in the Budget and elaborated in the March 2026 Rajya Sabha reply, but implementation framework is under finalization. Similar schemes (PRIP, PLI) typically release guidelines within 3–6 months of announcement.
Step 4: Build a Strong Proposal Focus on:
Step 5: Submit When Portal Opens (Anticipated Process) Once guidelines are released:
Kiran Mazumdar Shaw (Chairperson, Biocon Group): Described it as a decisive investment in India's health and innovation future, positioning the country as a global biopharma manufacturing hub through scale-up, regulation, new NIPERs, and clinical trials network.
Satish Reddy (Chairman, Dr. Reddy’s Laboratories): Called the ?10,000-crore Biopharma Shakti a key enabler for India's shift from volume-based generics leadership to value-driven global innovation in complex therapies.
Namit Joshi (Chairman, Pharmexcil): Hailed it as a historic shift to make India a biopharma powerhouse, building an end-to-end ecosystem for MSMEs in biologics/biosimilars, with SEZ/GST reforms, CDSCO strengthening, and NIPER expansion to boost exports and competitiveness.
Achin Gupta (MD & GCEO Designate, Cipla; Co-Chair, FICCI Pharma Committee): Noted healthcare's central role in Viksit Bharat, with emphasis on long-term capacity building in high-end manufacturing, advanced therapies, and self-reliance via Biopharma Shakti.
Shreehas Tambe (CEO & MD, Biocon Biologics): Viewed it as a much-needed step (with prior R&D commitments) to strengthen biopharma capabilities, innovation, advanced manufacturing, CDSCO cadre, NIPERs, and trial sites for affordable complex therapies.
Sheetal Arora (Promoter & CEO, Mankind Pharma): Highlighted biopharma's central place in manufacturing wave, with Biopharma Shakti supporting domestic ecosystem, global standards CDSCO, faster approvals, and patient access via duty exemptions.
Meenakshi Nevatia (Country President, Pfizer India): Welcomed Biopharma Shakti for scaling high-quality production in India, boosting confidence for R&D/manufacturing, and patient-centric measures like cancer drug duty exemptions amid NCD burden.
Dr. K. Anand Kumar (MD, Indian Immunologicals): Saw it as defining for vaccines/biologics hub transition, with investments in infrastructure, raw materials, and resilience to move from volume to innovation-led leadership.
Ashok Nair (MD, RPG Life Sciences): Noted practical focus on infrastructure (new/upgraded NIPERs, 1,000+ trial sites, CDSCO global standards) to accelerate biosimilars competition and market entry.
Nitin Jain (Founder and MD, Iberia Pharmaceuticals): Praised forward-looking steps in NIPERs, trial network, CDSCO, and domestic manufacturing to boost innovation in biologics/biosimilars and address NCDs.
Dr. Saloni Wagh (MD, Supriya Lifescience): Described it as mature policy for sustained investment in research, trials, and manufacturing depth to transition to innovation-led biopharma leadership.
Sanjay Vyas (President, MD, GCC Head India, Parexel): Called the 1,000 trial sites pivotal for improving research quality, speed, and global appeal in clinical development.
Rajwinder Mehdwan (MD & CEO, Roche Pharma India): Welcomed healthcare centrality, Biopharma Shakti, CDSCO global standards, NIPERs, trial sites, and patient measures for value-led system and competitiveness.
Shweta Rai (MD – India, Bayer Pharmaceuticals): Appreciated biopharma strengthening, R&D incentives, duty rationalisation, trial sites, CDSCO enhancements, and patient access for value-driven leadership.
The biopharma sector faces high entry barriers, such as complex manufacturing processes, cold chain logistics, and stringent regulatory oversight. The 10,000 crore Biopharma SHAKTI Scheme addresses these challenges by providing dedicated funding, infrastructure, and regulatory support from the government. MSMEs can navigate these difficulties by starting with upstream components like raw materials or intermediate products, bit by bit steadily scaling to full biologics manufacturing as they build capacity and expertise. This phased approach reduces some major risk and enables long-term sustainability.
The Rs 10,000 crore Biopharma SHAKTI Scheme is more than just a funding program—it represents India’s bold move into the global biologics landscape. With the country’s pharma exports already standing at a remarkable USD 30.47 billion and the biopharma market poised for an 8.3 percent CAGR, this scheme offers an unparalleled opportunity for growth. Companies that prepare now will not only secure grants and equity but also gain access to world-class infrastructure and expedited regulatory support.
While we’re still waiting for the official application portal to open, now is the time to get ahead. Start by proactively working on your registration, documentation, and proposal development. Stay updated by regularly checking pharma-dept.gov.in and PIB for the official notification. Once it arrives, speed will be key—acting swiftly could be your ticket to taking full advantage of this transformative scheme.
As I was writing this piece, I couldn't help but feel the weight of the transformation unfolding before us. It's more than just an opportunity—it’s a moment in time when we, as a nation, stand at the cusp of something groundbreaking. The Biopharma SHAKTI Scheme isn’t merely about securing funding; it’s about paving the way for homegrown innovation to thrive on the global stage.
This is our time to not only grow but to lead, to push boundaries, and to prove that India is not just following the global biologics wave but is setting the pace. Witnessing this shift, knowing we are part of it, feels like being at the beginning of something extraordinary.
Is the scheme open only to large companies?
No, startups and MSMEs are explicitly encouraged, especially in innovation and component manufacturing.
Will there be equity funding?
Yes, structured equity support is a core component for taking candidates through development milestones.
How does it differ from PRIP or PLI?
PRIP and PLI focus more on chemical entities and production incentives; SHAKTI is biologics-specific with heavy emphasis on clinical trials, regulation, and human capital.
Can foreign companies apply?
Likely through Indian subsidiaries or joint ventures with majority Indian ownership, subject to final guidelines.